Introduction
Behind every progressive kidney disease lies an alteration at the cellular level- human kidney fibroblasts are currently shaping future renal research and regenerative medicine.
Human Kidney Fibroblast, or ‘renal fibroblast’, plays a primary role in the synthesis of collagen and extracellular matrix (ECM). The cells are thought to be a key factor involved in renal fibrogenesis. Globally, the burden of kidney disease is more than 850 million. This reflects the importance of kidney research focused on understanding cellular drivers of fibrosis. The kidney fibroblast cell line plays a crucial role in developing advanced cellular models. This helps in unveiling the underlying cellular and development of advanced treatment in renal disease.
What Are Human Kidney Fibroblasts?
Human kidney fibroblasts are mesenchymal cells that have a spindle-shaped morphology. The cells are found in the renal cortical interstitium and maintain ECM homeostasis. The major structural and functional role of the human kidney fibroblast includes:
- Foundational framework for renal cortical interstitium
- Contributes in synthesis of Collagen I and ECM
- Synthesis of erythropoietin, a glycoprotein that controls red blood cell production
- Produces prostaglandins, a hormone that regulates electrolyte and water balance in the body
- On injury, the transdifferentiation of cells into myofibroblasts, which results in excessive matrix deposition (fibrosis)
- In kidney injury, human kidney fibroblast forms tertiary lymphoid tissue, promoting chronic inflammation.
Human Kidney Fibroblasts in Kidney Fibrosis Research
In kidney fibrosis research, Kidney fibroblast cell lines are a crucial model for researchers in renal studies. The major research application includes:
Mechanism of Fibroblast Activation
- Fibroblast plays a crucial role in wound healing, fibrosis, and cancer development.
- All the functional aspects occur with fibroblast activation.
- A complex process that involves dynamic phenotypic changes, such as alteration of cellular morphology, contractile property acquisition, or cytoskeletal organization
- The activation is believed to be a fibroblast-to-myofibroblast transition (FMT) [2]
Myofibroblast Transition and α-SMA Expression
- FMT transition occurs in the form of three cell types- quiescent, proto-myofibroblasts, and myofibroblasts
- Functional role evolves from quiescent state to secretory phenotype.
- Phenotypic alternation indicated with α-SMA expression upregulation (Figure 1) [2]
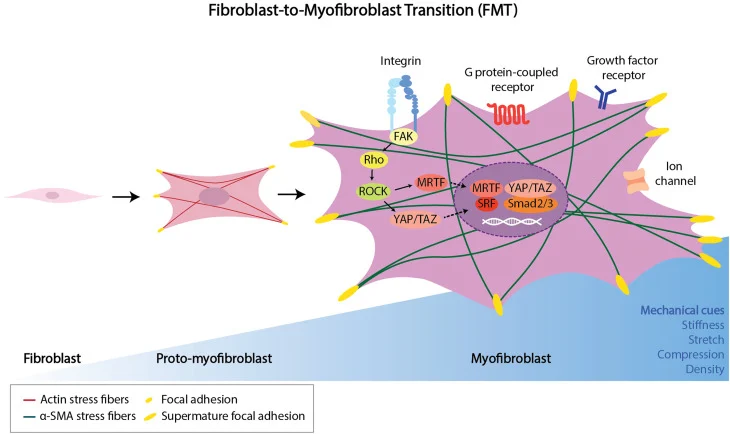
Illustration of FMT
Excess ECM Deposition and Scarring
- Enable researchers to understand the dynamic relationship between ECM deposition and fibroblast activation.
- Applicable in wound healing studies, it indicates the fibroblast’s role in tissue integrity restoration.
- Understanding the mechanobiology of human kidney fibroblast and the development of targeted medicine
- Fibrosis-encapsulated tumoroid (FET) model enables fibroblast encapsulation, forms fibroblastic shell (scarring) via ECM deposition
Contributes to Chronic Kidney Disease (CKD) Progression
- Kidney fibroblast contributes to scarring, a major contributor to CKD.
- Kidney Fibrosis Research enables understanding of phenotypic changes and ECM production.
- Aid in the development strategies of disease reprogramming
Kidney Fibroblast Cell Line: Applications in Laboratory Research
Common Kidney Fibroblast Cell Line
- NRK-49F (Normal Rat Kidney)
- PK-15 (Swine)
- HKF (Human Kidney Fibroblast)
- MES13 (Mouse)
Primary vs Immortalized Kidney Fibroblast Cell Line
- Human Kidney fibroblast categorised as: primary cells and immortalised cell lines.
- Primary human kidney fibroblasts, isolated from renal tissue, mimic human physiology, maintain native genes, understand ECM secretion patterns, and cytokine responsiveness. However, cells have limited lifespan and donor variability.
- Immortalised kidney fibroblast cell lines, genetically modified (e.g., via hTERT or SV40 transformation). Exhibits slight phenotypic drift, provide stable and reproducible model suitable for long-term mechanistic studies, facilitates high-throughput assays, and pathway analysis in renal research
Advantages in In-Vitro Disease Modeling
- Model for renal fibrogenesis
- Stimulated by pro-fibrotic factors (TGF-β1)
- differentiate into myofibroblast-like phenotypes characterized by α-SMA expression
- Increased α-SMA expression and collagen I/III
- ECM deposition, fibroblast activation pathways, epithelial–mesenchymal crosstalk, and inflammatory signalling
- Advanced 3D cultures and co-culture systems
Drug Screening and Anti-Fibrotic Compound Testing
- Study drug action in targeting various molecular signalling pathways (TGF-β/SMAD, Wnt/β-catenin, NF-κB)
- Response in collagen secretion, fibronectin expression, ROS production, etc.
- Serves as a preclinical renal research platform
Reproducibility and Scalability in Research
- Kidney Fibroblast Cell Line offers high reproducibility.
- Enables result replication
- Essential for translational nephrology and drug development
Role in Injury Repair and Regenerative Mechanisms
Response to Acute Kidney Injury (AKI)
Renal fibroblasts become rapidly activated in response to hypoxia, oxidative stress, or inflammatory response from damaged epithelial cells. Fibroblasts contribute to tissue stabilization, producing extracellular matrix (ECM) components, maintains structural integrity. Participates in wound-healing signalling cascades (TGF-β, PDGF, and FGF pathways). Persistent activation contributes to interstitial fibrosis—progress towards chronic kidney disease (CKD).
Secretion of Growth Factors and Cytokines
Fibroblast activation contributes to paracrine signalling. Secretes growth factors (HGF, VEGF, TGF-β, CTGF). Releases pro-inflammatory cytokines (IL-6, TNF-α, and MCP-1), that regulates recruitment of immune cells. Contributes to tubular regeneration, angiogenesis, and ECM remodelling.
Potential Targets for Regenerative Modulation
Regenerative research to understand the mode of fibroblast activation. Development of anti-TGF-β inhibitors, epigenetic modulators, exosome-modulated signalling, etc. Targeting fibroblast plasticity underlays a promising direction in regenerative renal medicine.
Role of Human Kidney Cells Interaction Network
The major research areas include the study of:
- Fibroblast–epithelial interaction
- Understanding Crosstalk with Immune Cells
- Endothelial signalling and vascular remodelling
- Detailed study in translational nephrology
Emerging Innovations in Regenerative Renal Research
Stem Cell–Derived Fibroblast Studies
- Mesenchymal stem cells (MSCs) differentiate into specific cell types, including renal stromal and fibroblast-like cells.
- Contributes to understanding fibroblast lineage specification and plasticity
- Detailed understanding of molecular pathways (fibroblast activation, ECM remodelling, and myofibroblast transition)
- Stem cell–derived systems also enable patient-specific disease modelling, offering insight into genetic susceptibility to renal fibrosis.
Exosome-Based Fibroblast Activity Modulation
- Stem cells–derived exosomes act as cargo molecules. Exosomes carry essential proteins, microRNAs, growth factors, and anti-inflammatory cytokines, potentially reducing FMT.
- Exosome therapy offers a cell-free regenerative strategy.
- Current research evaluates the role of ECM deposition and reverse fibrotic changes.
Precision Medicine Approaches
- Integration of transcriptomics and proteomics identifies fibroblast-specific biomarkers and molecular signatures involved in kidney fibrosis.
- Development of CRISPR-based gene editing tools
- Development of personalised therapeutic strategies targets patient-specific fibrotic pathways.
- Improvement in treatment efficacy minimises adverse events.
Therapeutic Implications and Future Directions in Kidney Fibrosis Research
There is a wide scope of use of human kidney fibroblast in fibrosis research. This includes:
- Development of anti-fibrotic therapy
- Identification of Biomarker
- Personalized renal regenerative strategies
- Addressing clinical translation challenges
Conclusion
Human kidney fibroblast cells have redefined the field of renal research. It contributes to providing structural support to the development of regenerative potential. The kidney cell line contributes to the development of next-generation kidney therapies. Research implication contributes to future kidney fibrosis research.
References
- International Society of Nephrology. More than 850 million worldwide have some form of kidney disease: help raise awareness. 2025. Available from: https://www.theisn.org/more-than-850-million-worldwide-have-some-form-of-kidney-disease-help-raise-awareness/
- Chang Y, Lee JW, Holle AW. The mechanobiology of fibroblast activation in disease. APL bioengineering. 2025 Jun 1;9(2).
FAQ’s
Q- What are Human Kidney Fibroblasts?
Human kidney fibroblasts are isolated from human kidney tissue. These cells play an important role in the synthesis of collagen I and ECM.
Q- What is the Importance of Kidney Fibrosis Research?
Kidney disease contributes widely to the global health burden. The treatment of its severe form is challenging. The use of human kidney fibroblasts plays a crucial role in kidney fibrosis research.
Q- How Does Fibroblast Lead to Severe Kidney Disease?
Fibroblast plays a crucial role in tissue injury repair mechanisms. The damage to kidney cells leads to fibroblast activation. This leads to excessive deposition of ECM at the site of damage.
Q- However, Do Human Kidney Fibroblasts Contribute to Regenerative Research?
Researchers have explored fibroblast cells in the area of fibrosis modelling, repair mechanisms, cell-to-cell communication, targeting regenerative modulation, etc. This model contributes to understanding renal healing.