Introduction
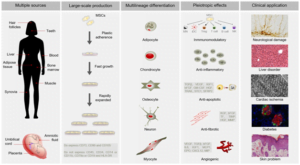
Advancements in regenerative medicine research have positioned adipose-derived stem cells (ADSCs) as a critical tool. ADSCs are the subset of mesenchymal stem cells (MSCs). These cells are abundant in the body (adipose tissue/ fat cells). The cells possess multi-lineage plasticity and deliver active regenerative potential. These cells influence immunomodulation, angiogenesis, tissue remodelling, and anti-inflammation at the molecular level. Regenerative medicine is vital to healthcare research and therapeutics, potentially delivering cures for chronic, untreatable diseases and conditions.
The in-depth study of human adipose-derived stem cells provides deeper insights into the mechanism governing the healing process.
Biological Characteristics of Human Adipose-Derived Stem Cells
Adipose-derived stem cells are isolated from adipose/fat cells from specific areas of the body. The cells are multi-lineage, that is, they can differentiate into adipogenic, osteogenic, chondrogenic and neural cells. The key characteristics of human adipose-derived stem cells (hADSCs) include:
Cellular Morphology
- hADSCs exhibit fibroblast-like spindle-shaped morphology
- adheres to the plastic-coated surface
- forms a homogenous monolayer at early passage
- displays contact inhibition and displays an organised alignment pattern
- Prolonged time in culture pushes cells towards senescence-associated changes.
- Morphological changes include an enlarged, flattened cell structure.
Surface Markers (Immunophenotype)
- Characterised by a cluster of differentiation (CD) markers
- Positive markers: CD73, CD90, and CD105 (MSCs markers)
- Negative markers: CD34, CD45, CD14, CD19, and HLA-DR (hematopoietic & endothelial markers absent)
Differentiation potential (Multipotency)
ADSCs differentiate into distinct lineages under specific conditions
- Adipogenic lineage (fat cells)
- Osteogenic lineage (bone-forming osteoblasts)
- Chondrogenic lineage (cartilage-producing chondrocytes)
- Other lineage: Neurogenic, endothelial-like phenotype, myogenic
Isolation Procedure of Human Adipose-Derived Stem Cells
The isolation of hADSCs follows a multi-step process. This includes:
- Source: Visceral fat or subcutaneous tissue
- Tissue Collection & Washing: Tissue is collected using needle biopsy or liposuction. The tissue is washed with 1X phosphate buffer saline.
- Enzymatic Digestion: Collagenase Type 1 (0.75-1.0%) are used for tissue digestion. The condition maintained includes 37°C for 30-60 minutes in a shaker to release stem cells from the extracellular matrix.
- Neutralisation: A complete culture medium is used for neutralising collagenase
- Centrifugation: Centrifuged at 300-500 g for 5-10 min
- SVF Purification: Supernatants are discarded. The cell pellet consists of the stromal vascular fraction. It is washed and resuspended in a complete culture medium.
- Filtration: The cell suspension is passed through mesh (70-100 μm). This removes debris, and cells are plated in coated culture flasks and incubated in CO2 incubators. The environment was maintained at 37°C, 5% CO2, and a relative humidity of 50%.
Mechanisms of Action in Regenerative Research
Self-Renewal Capacity
- High proliferative rate in comparison with other sources, such as bone marrow
- Maintains self-renewal capacity
Paracrine Signalling and Secretion of Growth Factors
- Release secretome- cytokines, growth factors, chemokines and extracellular vesicles
- Active signalling molecules released- VEGF, FGF, TGF-β, HGF
- Major role in angiogenesis, tissue repair, and anti-apoptotic signalling
Immunomodulation
- Suppress T-cell proliferation
- Modulates macrophage polarization
- Regulates inflammatory cytokines
- Suitable for application in autoimmune disease

Promote Angiogenesis
- Enable neovascularisation, secretes pro-angiogenic factors
- Interacts with endothelial cells
- Promote ischemic tissue repair
- Aids in the wound healing mechanism
Genetic Stability & Senescence
- Maintain genetic stability in a controlled laboratory environment
- Prolonged expansion can lead to replicative senescence, DNA damage accumulation, and other effects.
- Identification of senescence markers and karyotyping is essential to ensure cell quality
Metabolic Activity and Adaptability
- hADSCs exhibit metabolic flexibility
- Shifts between glycolysis and oxidative phosphorylation based on environmental conditions
- Survives in adverse microenvironments (hypoxic or injury-related microenvironments)
What are the Applications of hADSCs?
Human adipose-derived mesenchymal stem cells have a wide range of applications. Regenerative medicine is explored in various chronic diseases, including:
- Orthopedic and musculoskeletal/bone regeneration
- Anti-aging and skin rejuvenation
- Wound healing, Scar removal
- Cardiovascular and ischemic conditions
- Neurodegenerative disease (ALS, MS, MND, Autism)
- Spinal cord injury
- Hair rejuvenation in conditions like alopecia aerata, androgenic alopecia, etc.
Clinical Evidence for the use of hADSCs in Chronic Diseases
Osteoarthritis
- In the Phase IIb clinical trial, there was a significant improvement in the WOMAC pain and function score
- Cartilage repair and restoration of synovial fluids
- Sustained pain relief and improvement in joint mobility up to 2 years [1]
Diabetic Foot Ulcer
- Faster wound closure
- Significant wound healing in comparison with the control group [2]
Critical Limb Ischemia
- Improved blood flow and angiogenesis
- Improved ankle-brachial index (ABI)
- Reduced ulcer severity and pain management [3]
Neurodegenerative Disease
- Improvement in spinal cord injury
- Reduction of inflammatory biomarkers, reduced inflammation
Wound Management (Non-Diabetic)
- Rapid re-epithelialization
- Increased collagen production and deposition
*NOTE: The studies reported the safety profile, tolerance and efficacy of the regenerative medicine. No major side effects were reported.
What are the Advantages of hADSCs over Other Sources?
Adipose-derived stem cells have advantages in comparison with other sources, including:
- Abundance and ease of harvest
- The collection procedure is minimally invasive
- The collection procedure is relatively easy
- Higher yield of stem cells and high proliferation capacity
- Clinical scalability and ease in research adaptability
What are the Safety Concerns in Using hADSCs?
Currently, the research is at an early stage. The major concerns include:
- Quality of the adipose-derived stem cells for research purposes and clinical application
- Standardisation of isolation and culture conditions
- Cell heterogeneity based on the source potentially displays non-replicative results
- Research translation from the lab to clinical use is efficient
- Marker expression varies based on donor variability, isolation technique, and culture conditions.
*Kosheeka, India, is a leading GMC-certified stem cell research laboratory. The organisation supplies various stem cells isolated for research purposes
What are the Emerging Trends & Future Directions?
- Advancement in developing cell-free therapies (exosomes)
- Development of personalised regenerative medicine
- Development of combinational therapy (combining regenerative medicine with conventional treatment)
- Large-scale clinical trials for establishing efficacy, safety, and tolerance among the patients
Conclusion
Adipose-derived stem cells differentiate into multiple lineages. It broadens the scope of use in regenerative medicine. hADSCs are abundant in the body, making them easy to procure. Preclinical and clinical studies have marked the tolerance, safety and efficacy of regenerative medicine in various chronic diseases. Currently, there is an immense need for thorough research and for transforming regenerative medicine into a therapeutic application.
References
- Lee S, Chae DS, Song BW, Lim S, Kim SW, Kim IK, Hwang KC. ADSC-based cell therapies for musculoskeletal disorders: a review of recent clinical trials. International Journal of Molecular Sciences. 2021 Sep 30;22(19):10586.
- Holm JS, Toyserkani NM, Sorensen JA. Adipose-derived stem cells for treatment of chronic ulcers: current status. Stem cell research & therapy. 2018 May 15;9(1):142.
- Chan TM, Harn HJ, Lin HP, Chiu SC, Lin PC, Wang HI, Ho LI, Chuu CP, Chiou TW, Hsieh AC, Chen YW. The use of ADSCs as a treatment for chronic stroke. Cell transplantation. 2014 May;23(4-5):541-7.
FAQ’s
Q- What are ADSCs?
ADSCs are mesenchymal stem cells. The cells are isolated from adipose/fat tissue. These cells are multilineage, that is, they can regenerate into multiple cell types in response to external stimuli.
Q- Are Adipose-Derived Stem Cells Safe for Clinical Use?
Preclinical and clinical trials have marked the safety and tolerance of ADSCs. The studies are still at an early stage. There is a need for large-scale clinical trials to establish long-term safety and efficacy.
Q- What Chronic Diseases are Studied Using hADSCs?
Various diseases, including neurodegenerative conditions, osteoarthritis, musculoskeletal conditions, CLI, Crohn disease, diabetes and so on.