“There is no good science with the bad models of toxicological analysis” the statement may sound quite apodictic, but it is apt to the today’s situation, wherein animal models are completely banned. In this regard, a good model should conveniently offer the best results in the context of accuracy, reproducibility, and reliability. Depending upon the task that is required to be performed there are both simple as well as complex models; although simple models are convenient to use and can offer more reproducible results, the issue is always to be fit for the purpose.
Today’s pharmaceutical industry is struggling with many inherent toxicological issues that are complex in nature, involving a larger network of in vivo biological system. Thus, it is always better to be closer to what our system is; in order to obtain more relevant results. In order to ensure complete safety as well as reliability of the results, formal validation with different models and their comparative analysis is the key to success.
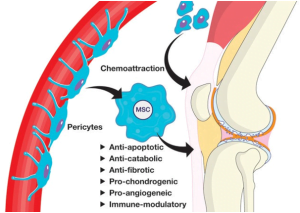
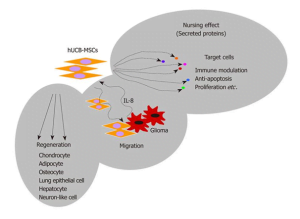
In this regard, cells derived from human primary tissues are becoming more and more popular in recent years, especially in the preclinical analysis of drugs, toxic compounds, etc. While the use of animals is almost on the verge of being stopped, the field of life sciences and other research has reached towards the transition into completely new research and development tool. During the last couple of decades, the industry has experienced an exponential increase in the advancements of technologies required to maintain viable cultures for a longer period of time to enable their study and testing applications. However, until recently the access to human primary cells was very limited but human stem cells and their potential to differentiate into various cells of other lineages become available; and is further impacting relevant research in the life science industry including toxicological studies.
In this regard, the shortcomings of the traditional cultures and the application of stem cells in the industry are being discussed further.
Shortcomings of traditional methods in cell culture
Sometimes, the art of cell culture may often lead to the artifacts of cell culture; due to the requirement of a number of stringent parameters. However, there are few limitations in the process that can further be discussed herewith:
- Cell sources
It has always been very difficult to obtain primary cells from human tissues in large quantity and quality; with very few exceptional tissues like human blood, placenta, umbilical cord, etc. However, it is very important to understand that due to the involvement of different regulatory authorities, it is always easy to obtain primary tissues for extraction of cells for pharmaceutical analysis. However, the field is still struggling with certain limitations like cost, quantity as well as inter-individual differences.
- Cell culture contamination
Contamination is the main issue that is being tackled by the experts since a longer period of time. Since, contaminations can have a severe impact on cellular production, cellular transformation, and genetic instability; the entire scientific world is still struggling with minimizing their attack on cellular culture.
- Cellular differentiation and organ functionality
The genetic expression of the cellular phenotype is much more dependent upon the extent of gene expressions. For many of the applications related to toxicological studies, it is more convenient to use cells that are terminally differentiated. For example, stem cells that are being used in the in vitro analysis, it is very important to maintain them in a pluripotent state.
Thus, the many problems that are being encountered in traditional cell culture can be controlled by maintaining cells in good physiological conditions and setting up ideal parameters.