According to research published in 2015 in BioMed Research International by Florencio-Silva and team, apart from protecting the body, the bones are involved in locomotion, containing the vital bone marrow as well as storing calcium and phosphate. While it looks inanimate, the bone shows dynamic remodelling with bone resorption and formation performed by cells called osteoclasts and osteoblasts, respectively. The precise remodelling is important for calcium homeostasis and repair of fractures. A loss of balance between the formation and resorption results in bone diseases such as osteoporosis if the resorption is more or osteopetrosis if the formation is more.
4–6% of the total resident bone cells are called osteoblasts: the bone- forming cells. These cells are derived from mesenchymal stem cells (MSC). Various aspects of how these cells form the bone and the processes that regulate these cells require the in vitro culture of osteoblasts. The use of cell cultures has allowed scientists to study important mechanisms associated with the functions, division, survival and differentiation of osteoblasts.
Several articles describe the isolation and culture of primary osteoblasts from many sites of the body from rats, mice and humans. Despite the difference in locations, similar behaviour of cells is seen in vitro. Culturing calvarial osteoblasts from neonatal rats can allow studies of the functions of these cells as the process is easy and yields large amounts of cells.
Osteoblasts isolated from calvarial and long-bones can allow quantification of the bone formation by the cells. Additionally, the cells can be traced from their immature stages to their mature stages. The extracellular environment can be controlled in cultures giving these primary cultures an edge over in vivo studies. Thus, primary osteoblast culture emerges as a key player in bone research.
A team led by Taylor reported key points to isolate osteoblasts thereby allowing various studies into the cell functions and bone diseases. The points include:
- For human bones, explant cultures can be established from osteoarthritis patients scheduled for total shoulder replacement surgery with ethical approval. As aged or diseases samples (mouse/rat/ human) contain more fat and marrow, the removal of these tissues is important to prevent contamination by their cells.
- The recommended seeding densities are 105cells per well for 6-well plates; 5 × 104 cells for 12-well plates and 2.5 × 104for 24-well plates.
- Cell dissociation using Accutase is gentler than trypsin-EDTA to thus lower the issue of non-viable cells.
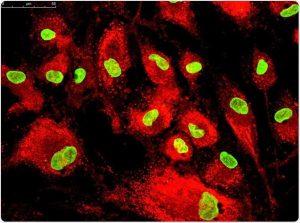
- Confluency is seen in rodent cultures by day 4 of culture. The deposition and mineralization of the bone can be seen under an electron microscope.
- Bone nodules form in 3-weeks for human samples isolated from <40 years old patients. The age, sex, race and disease status influences the bone cultures.
- Maintenance of the pH at 7.4 is vital as changes can affect mineralization.
- αMEM and DMEM can be used for rat osteoblasts while αMEM is apt for culturing mouse osteoblasts.
- As the formation of bone nodules requires a source of Phosphate, β-glycerophosphate is used in vitro; appropriate and low levels are important (2 mm).
References:
Rinaldo Florencio-Silva, Gisela Rodrigues da Silva Sasso, Estela Sasso-Cerri, Manuel Jesus Simões, and Paulo Sérgio Cerri. Biology of Bone Tissue: Structure, Function, and Factors That Influence Bone Cells. BioMed Research International 2015; Article ID 421746: 17 pages.
Taylor S E, Shah M and Orriss I R .Generation of rodent and human osteoblasts. BoneKEy reports 2014; 3: 585.