Introduction
Human dermal fibroblast (HDFs) produce an extracellular matrix (ECM). They form a connecting matrix that forms connective tissues and have a crucial role in wound healing. Human dermal fibroblast cells are an excellent model that helps researchers to study various aspects of cellular physiology, drug testing, toxicology, skin biology, and regenerative medicine.
The application of Human Dermal Fibroblast Cells is widespread. Dive deeper into understanding the elaborate application of these cells in biomedical research.
Biological Characteristics of Human Dermal Fibroblasts
Historical Perspective of HDFs
- HDFs in the research field progressed over time as follows:
- In the 19th century, Rudolf Virchow described connective tissues as ‘spindle-shaped cells’
- In the 20th century, the demonstration of fibroblast cell growth in in-vitro condition
- In the late 20th century and 21st century, the Human Dermal Fibroblast Cell Line developed. Based on its heterogeneity, several subtypes were obtained, revolutionizing regenerative medicine research
Cellular Morphology and Phenotypic Markers
- The morphological characteristics of human fibroblast cells vary based on their cell type. Some popular markers include- CD90, Vimentin, α-SMA, COL1A1, Fibroblast Surface Proteins TE-7, 1B10, etc.
- The type of biomarker studied depends on the experimental setup and purpose
Role in Extracellular Matrix Synthesis
Synthesis and regulation of ECM components like elastin, collagen, laminins, fibronectin, nidogens, and tenascin
- ECM turnover, water retention, and maintains cellular integrity
- In dermis, HDFs maintain tissue architecture activates wound healing process.
- Exhibits immunoregulatory functions through inflammatory secretome (TGFβ, chemokines, prostaglandins, interleukins), and influences immune activities.
- Expresses toll-like receptors, defends against microbial attack, and produces defensins
Fibroblast Heterogeneity
- Diverse roles in developmental tissue, maintenance, and disease
- Inter-organ heterogeneity in different anatomical locations
- Primary fibroblast cells originate during embryogenesis.
- Subtypes developed- quiescent fibroblasts (ECM homeostasis, supportive tissue function), dermal fibroblasts (skin tissues), Fibrosis-associated fibroblasts (chronic scarring and fibrogenesis), Cancer-associated fibroblasts (present in solid tumours), Aging-associated fibroblasts (obtained from aging tissue/environmental stress)
Fibroblast Cell Line: Source and Classification
Human Dermal Fibroblast Cell Line (HDFa). Originating from skin tissue, it is the primary cell line used for research applications.
Human Dermal Fibroblast ATCC have following details:
- Product Type: Human Cells
- Organism: Human
- Product Type: Primary Cells
- Tissue Origin: Skin
- Morphology: Spindle-shaped cells, bipolar and refractile
- Cell Type: Adherent
- Age: Adult
- Medium for Growth: Fibroblast Basal Medium
We understand that procuring a cell line from ATCC is hectic due to various factors like cost, complexity in regulatory barriers, logistics delays, challenges with cold chain maintenance, etc.
Kosheeka, Noida, India, is one of the GMP-certified laboratories that conducts cutting-edge research in biomedical science. Kosheeka is also a leading provider of high-quality primary and secondary cells for research purposes all over India.
Culture Conditions and Experimental Handling
Once the cells are received, one must follow the underlying procedure:
Unpacking and Culture
- Check for any leakage or breakage of the pack.
- Remove the cells from ice packing and immediately store at -130°C (liquid nitrogen)
- Prepare complete medium consisting of 10% FBS and 1% antibiotics (Penicillin-Streptomycin-Amphotericin B Solution)
- Add 5 ml complete medium to a T25 flask, pre-equilibrate the medium at 37°C, and place it in a CO2 incubator with 5% and adequate humidification.
- Culture vial gently agitated and thawed in a 37°C water bath
- Decontaminate the vial at 90% thaw stage and add culture in 1 ml complete medium
- Seed the cells in pre-equilibrated flasks and incubate at 37°C, in 5% CO2 incubator for 24 hours
Subculturing
- On reaching 80%-85% confluency passage the fibroblast cells
- Carefully aspirate the media from the flask
- Rinse the cells and flask with 2 ml 1X PBS per 25 cm2
- Trypsinize the cells with 1ml warm trypsin (37°C), and gently rock the flask to cover the whole surface
- After 2-3 min, add complete medium three times the volume of trypsin, avoiding over-trypsinization
- Aspirate all dissociated cells in a sterile centrifugation tube, centrifuge cells at 150 x g for 2 to 4 minutes.
- Aspirate out the suspension, add pre-warm 2 to 10 ml complete medium to the pellet and resuspend it.
- Seed 2500 to 4000 cells/cm2 of the surface area
- Flask gains confluence in 24-48 hours, and cells are used for other experimental processes
Precautions
- To avoid over-trypsinization of cells, which hampers cellular morphology and integrity, cells can reach early senescence, enter the early apoptosis pathway.
- Trypsinize cells at 80% confluency stage, to avoid overconfluency, which leads to differentiation, difficulty in trypsinization, and loss of morphological and cellular characteristics.
Role of Human Dermal Fibroblasts in Skin Physiology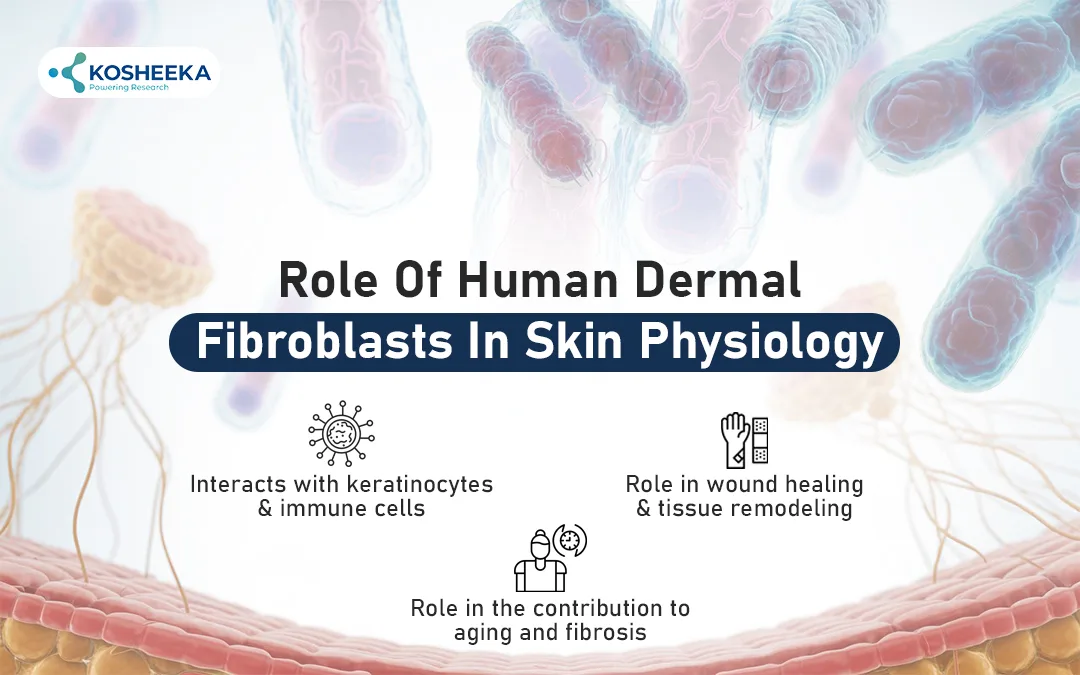
Interacts with Keratinocytes and Immune Cells
- Secrete growth factors (KGF, TGF-β, FGF) regulate keratinocyte proliferation, differentiation and homeostasis.
- Favours paracrine signalling, maintains epidermal barrier integrity, and skin architecture.
- Interacts with immune cells (macrophages, T cells, mast cells), releases cytokines and chemokines, and modulates immune response.
- Immune surveillance modulates inflammation.
- Dysregulated fibroblasts contribute to skin-associated chronic inflammatory conditions
Role in Wound Healing and Tissue Remodeling
- Fibroblasts migrates at wound site, differentiate into myofibroblasts (TGF-β signaling)
- Synthesize ECM components: collagen, elastin, fibronectin
- Regulates angiogenesis, secretes VEGF and other pro-angiogenic factors
- Impaired fibroblast activities result in delayed healing, and hyperactivity leads to pathological scarring
Role in the Contribution to Aging and Fibrosis
- Ageing reduces proliferative capacity and collagen production
- Fibroblast cellular senescence alters the function and composition of the ECM, leading to loss of skin elasticity
- Aged fibroblast secretes pro-inflammatory markers, contributes in inflammation
- Hyperactivation is a hallmark of fibrotic disorders, and excessive collagen deposition
- Human Dermal Fibroblast Cell Line is crucial in developing anti-fibrotic and regenerative therapies
Applications of Human Dermal Fibroblast Cell Line
The application of human dermal fibroblast cells is widespread, including:
- Oncology and immunology research
- Cancer-associated fibroblasts and tumor microenvironment studies
- Cytotoxicity and toxicology testing
- Developing molecular diagnostics
- Safety testing and Skin science
- Regenerative science and skin remodeling
- Genetic and epigenetic disease modeling
Role in Regenerative Medicine and Tissue Engineering
Fibroblasts are Support Cells in Skin Substitutes.
- Dermal fibroblast provides biochemical and structural support, and produces ECM protein
- Promotes epidermal stratification, maturation and co-culturing with keratinocytes, acts as a skin substitute
- Mimics native skin architecture, improves graft integration.
Paracrine Signaling and ECM Scaffolding
- Fibroblast cells secretes growth factors (VEGF, FGF, TGF-β), regulates cellular proliferation, repair mechanisms, and angiogenesis
- Create balance between cellular regeneration, prevents excessive fibrosis
Fibrosis, Chronic Wounds, and Inflammatory Skin Disorders
- Dysregulation activates excessive ECM deposition, delayed wound healing, sustaining inflammation.
- In regenerative research, dermal fibroblasts aid in developing engineered skin constructs, aid in wound healing, and reduce inflammation.
Translational and Drug Discovery Applications
- High-throughput screening and toxicity testing
- Role in cosmetic and pharmaceutical research
- Predictive modeling for human skin responses
Limitations and Challenges in Fibroblast-Based Research
- Variability in donor (in primary cells) can have batch-to-batch differences
- Possible Replicative senescence and phenotype drift
- In-vitro research (cell line-based research) have lower risk of donor variability, less physiological relevance
- Currently, Need for advanced standardization
Future Directions and Emerging Technologies
- Analysis of a single-cell study and fibroblast subpopulation mapping
- Integration with induced pluripotent stem cells (iPSC) and bioengineered skin systems
- AI-driven modeling and precision dermatology
Conclusion
Human Dermal Fibroblast holds the primary position in biomedical research. They have fundamental roles in skin biology, structure, repair, and cellular communication. These cells regulate EXC integrity, wound healing, tissue modelling and inflammation. In regenerative medicine research, fibroblast cells helps in exploring subjects like aging, fibroblast, oncology, and regenerative medicines
FAQ’s
Q- What are Human Dermal Fibroblast Cells?
Human dermal fibroblast cells are found in the dermis, which is responsible for producing ECM components like elastin and collagen. These cells are essential for maintaining skin integrity, tissue remodeling, and wound healing.
Q- What are Human Dermal Fibroblast Cell Lines?
Human dermal fibroblast cell lines are a standardized population of fibroblast cells that are cultured under controlled laboratory conditions. The cell lines are an adequate source for conducting in-vitro studies.
Q- Can Human Dermal Fibroblast Cells be used for Regenerative Research?
Yes, in regenerative medicine, human fibroblast cells act as skin substitutes. They help in developing tissue engineering constructs and wound healing models. They secrete growth factors that help in building the ECM.